Ground Granulated Blast Furnace Slag Blended Concrete

D. K. Jain, Research Scholar, J. Prasad, Associate Professor, and A. K. Ahuja, Associate Professor, Department of Civil Engineering, I. I. T. Roorkee.
Cement concrete with Ordinary Portland Cement (OPC) continues to be the pre-eminent construction materials due to its commendable performance in terms of strength aspects, but durability of this is not satisfactory particularly when it is exposed to aggressive environment. Same time, present global environmental requirements suggest the civil engineers for reducing the consumption of OPC. Use of mineral admixtures like Blast Furnace Slag, Fly Ash and Silica Fume etc. in concrete may be a suitable solution in such situation.
Based on literature available, the present paper discusses the effect of GGBFS blending in concrete over its properties in fresh and hardened states. Factors affecting the hydration and strength development of blended concrete are presented. The present paper discusses the resistance of GGBFS blended concrete to Chloride attack, Sulphate attack, Carbonation, Aggregate-Silica reaction and Frost attack with factors influencing its performance. Pore refinement and secondary gel formation in the GGBFS blended concrete are the main reasons.
Introduction
OPC based concrete continues to be the pre-eminent construction materials for use in any type of civil engineering structures because of its easiness in construction, its satisfying performance in strength requirements, better durability in normal environment, in comparison to other construction materials like steel, timber etc but at the same time some problems are also associated with this. First is environmental pollution and large energy requirement in the production of OPC. Production of one tonne OPC required approximate 4.0 G Joule energy and produced approximate one tonne CO2 gas in the environment. At present the cement industries produced approximate 7% of total CO2 produced in the world, which is very alarming to our protective Ozone layer. Second problem is the lower durability in aggressive environment. Concrete with OPC, which performed, very well over a period of about 100 years in the normal environment showed substantial damage within a few years of construction in the aggressive environment. Use of mineral admixtures like Ground Granulated Blast Furnace Slag (GGBFS), Silica Fume (SF), Fly Ash (FA) etc. in concrete may be the better solution in above conditions.These admixtures also offer benefits with respect to the cost of concrete.Based on the information available in literature the performance of GGBFS in blended concrete, with respect to properties in fresh state, in strength development and in durability aspects are discussed in the present paper. Durability aspects with reference to Chloride resistance, Sulphate resistance, Aggregate-silica reaction and Frost resistance are described. Hydration process of GGBFS and its reactivity with OPC is also discussed. Effects of important factors on properties of blended concrete in fresh and hardened state are highlighted.
Ground Granulated Blast Furnace Slag
Blast Furnace Slag is a by product obtained in the manufacturing of Pig iron in the Blast furnace and is formed by the combination of earthy constituents of iron ore with lime stone flux. Quenching process of molten slag by water is converting it into a fine, granulated slag of whitish color. This granulated slag when finely ground and combined with OPC has been found to exhibit excellent cementitious properties. Glass particles of GGBFS are the active part and consist of Mono-silicate (Q0-type), like those in OPC clinker, which dissolve on activation by any medium. Glass content in GGBFS is normally more than 85% of total volume. Specific gravity of GGBFS is approximately 2.7-2.90, which is lower than of OPC. Bulk density of GGBFS is varying from 1200-1300 kg/m3. Normal chemical composition of Indian GGBFS is shown in Table 1. GGBFS is more closure to OPC in chemical composition in compare to other mineral admixtures.Hydration of GGBFS
Hydration products of GGBFS are poorly crystalline Calcium Silicate Hydrate broadly similar to that formed from hydration of OPC, but with lower Ca/Si ratio (Jimenez et al., 2003). Due to lower Ca/ Si ratio, these hydrates have more alkali retention capacity. Hydration products of GGBFS effectively fill up the pores and increase the strength and durability of concrete. GGBFS requires activation to initiate hydration and the availability of a medium for continuing the hydration process. Slag hydration can be activated by using alkalies, lime, sulphate etc (Chemically activation), or by fine grinding (Mechanically activation) or by increasing temperature of concrete (Thermal activation). Various alkalies activators like Sodium hydroxide, Sodium carbonate, Sodium sulphate, Sodium silicate (Water glass) etc. can be used for slag. Water glass activated slag produced most cross-linked structures that results in increased mechanical strength of hydration products, while Sodium hydroxide make hydration process of slag more intensive (Garcia et. al., 2003). Due to higher activation energy of blast furnace slag relative to OPC, it has advantage of thermal activation on its hydration (Roy and Idorn, 1982).| Table 1: Chemical Composition of GGBFS | ||
| Oxides | Quantity | Remark |
| SiO2 | 30 - 35% | - |
| Al2O3 | 8 - 22% | Higher in Indian Slag |
| CaO | 27 - 32% | Lower in Indian Slag |
| MgO | 7 - 9% | - |
| Fe2O3 | 8 - 10% | Higher in Indian Slag |
Reactivity of GGBFS with OPC
Hydration products like alkailies, lime and heat of OPC are activate the hydration of GGBFS particles in blended cement concrete. Initially and during early hydration of concrete containing GGBFS, the predominant reaction is with Alkalies hydroxide, but a subsequent reaction is with Calcium hydroxide (Roy and Idorn, 1982). Reactivity of GGBFS with OPC in blended concrete is depends on chemical & mineralogical composition, glass content, fineness etc. of the GGBFS. Type of portland cement employed and curing conditions also has a significant effect on the rate of formation of hydration products in blended concrete. On the basis of chemical composition of GGBFS various indices have been proposed in the literature to evaluate its reactivity Table 2. These formulas are not considering the effect of composition of OPC on hydration of blast furnace slag. Mantel (1994) reported the more reliability on first three formulas. Reactivity of high glass content slag is normally found more and greater fineness of slag also increases its reactivity due to increase in surface area for reaction with activators. Use of rapid hardening cement in place of OPC increases the reactivity of slag in blended concrete, due to more activators available at early age.| Table 2: Indices for Reactivity of GGBFS | ||
| Indices as per Chemical Composition | Recommended values | |
| CaO / SiO2 | 1.3 - 1.4 | |
| (CaO + MgO) / SiO2 | > 1.4 | |
| (CaO + MgO) / (SiO2 + Al2O3) | 1.0 - 1.3 | |
| (CaO + 0.56Al2O3 + 1.4MgO) / SiO2 | > 1.65 | |
| (CaO + MgO + Al2O3) / SiO2 | > 1.0 | |
ACI (ACI 233R-95) recommends the use of Slag Activity Index (SAI) to evaluate its reactivity. SAI is the percentage ratio of the average compressive strength of slag blended cement mortar cubes (at 50% slag content), to the average compressive strength of reference cement mortar cubes at a designated age. Based on SAI the GGBFS is classified into three grades namely, Grade 80, 100 and 120. Blended concrete with grade 120 normally achieved strength of OPC concrete at 3rd day and after, while concrete with grade 100 achieved at 7th day and afterward. However, concrete made with grade 80 GGBFS will have a lower strength at all ages and not recommended by ACI for use in structural concrete.
Mix Proportioning
No specific mix proportioning method is available for GGBFS blended concrete. Simplest and most common method of incorporating slag in concrete is a straight forward replacement of OPC by equal weight, but due to the difference in specific gravity of these two materials slight adjustment in aggregate content is needed for correct yield. Swami (1990) suggests that the total cementitious material has to be increased by 10% for 50% replacement and by 20% for 65% replacement level in blended concrete to attain a strength comparable to normal OPC concrete. Babu and Kumar (2000) used the efficiency concept for developing equal 28 days compressive strength for blended and normal concretes. They suggested the value of overall efficiency from 1.29 to 0.7 for 10 to 80% replacement level. Normally 30 to 50% replacement of cement by GGBFS is adopted in the field.Properties of Fresh Concrete
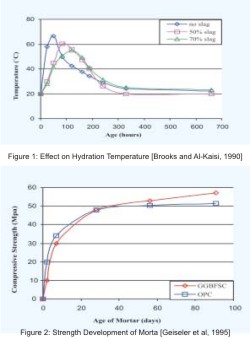
Strength Development
Strength development of GGBFS blended concrete is quite different from OPC concrete. Strength of blended concrete at early ages is lesser in compare to that of OPC concrete. Due to the prolonged hydration process of slag the later age strength of blended concrete is higher than OPC concrete. Results found by Geiseler et. al (1995) for the strength development of two equal grades mortars with and without GGBFS are shown in Figure 2. They also observed the 100% increase in strength of GGBFS blended mortar, after a period of 25 years, in compared to 28 day Strength.Strength development of blended concrete depends on properties of GGBFS, type of OPC, proportion of ingredients, and curing conditions. Olorunsogo and Wainwright (1998), studies the effects of particle size distribution of GGBFS on compressive strength of blended mortar. They use position parameter (x0) to serves as an indicator of the degree of fineness of slag and the slope of curve (n) to represents the size range of particles of different GGBFS. They found that the strength of mortar is reducing when value of position parameters (x0) is increasing for constant slope value (n), but effect of change in slope value (n) for constant position parameter (x0) is very little (Figure 3 a & b).
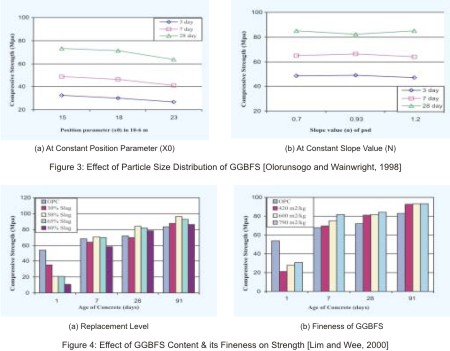
Lim and Wee (2000) and Jain and Pal (1998) present the effect of replacement percentage and fineness of GGBFS on strength development of blended concrete. They found that after the age of 28 days of moist curing, strength of 50, 65,and 80% GGBFS blended concrete are higher than the OPC concrete, while at 91 days blended concretes (with all replacement level) shows higher strength. Results of both studies show the optimum content of GGBFS equal to 50% for maximum strength. Blended concrete with higher fineness of GGBFS shows higher strength up to the age of 28 days, but at the age of 91 days strength of concrete with GGBFS of different fineness is very similar and significantly higher than the OPC concrete (fig 4 a & b). Therefore, toincrease the strength development at early ages finer grinding of GGBFS is useful.
Curing condition has much more effects on strength development of GGBFS blended concrete due to its slow and prolonged hydration. It required longer and continuous moist curing for complete hydration of slag particles. Sanjayan and Sioulas (2000), Miura and Iwaki (2000) and Brooks and Al-Kaisi (1990) studies the effects of curing conditions on strength of GGBFS blendedconcretes. They found that early age strength development (up to 28 days) of GGBFS blended concrete is increasing with curing temperature, but the effect on later strength is very little. Miura and Iwaki (2000) found that the effects of curing method on strength development are found only after age of 7 days. Blended concrete cured under water shows higher strength than sealed cured concrete. The detrimental effect of isolating specimens from a continuous supply of moisture where more on later age strength of GGBFS blended concrete. Temperature matched curing is also increases early age strength of concrete, particularly in the case of high GGBFS content concrete.
Chloride Resistance
Chloride resistance of concrete is the most important aspects for durability of RC structures. When chloride content in concrete reaches more than the threshold value, the protective layer of alkalinity get broken and steel reinforcement will corrode in the presence of oxygen and humidity. Hydration products of cementitious materials are also reacts with chloride and form Freidel's salt that does not have any harmful effects on concrete durability.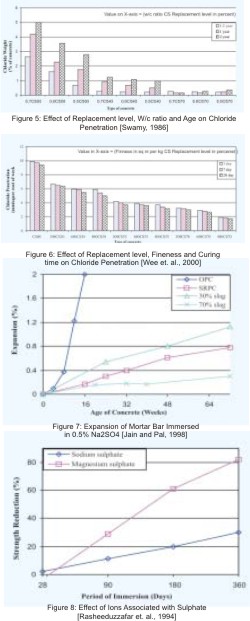
Kumar et. al. (2002) presents the results of depth of chloride penetration in concrete after 6 months exposure in 3.5 % Sodium chloride solution and found that penetration decreases with increase in GGBFS content. Corrosion studies of Jain and Pal (1998) show that corrosion resistance of blended concrete with 50% or above GGBFS gives real advantages. Swamy (1986) observed that in the first 10 mm layer, there was little difference in the amount of soluble chloride in OPC and GGBFS blended concrete, but at larger depth chloride content in GGBFS concrete were significantly lesser. Author reported that for a particular mix proportion the diffusion of chloride is reduced with decrease in the w/cm ratio or with increase in GGBFS content (Figure 5). Wee et. al. (2000) reported that the higher replacement level, higher fineness, and longer moist curing increase the chloride resistance of GGBFS blended concrete (Figure 6). Hope and Ip (1987), studies the corrosion of steel in blended concrete, after various cycles of wetting and drying in 3.5% sodium chloride solution and found that the corrosion of the steel is decreases with increase in GGBFS content. Al-Amoudi et. al. (1993) studied the long term (7 years) corrosi on resistance of concretes in 5% Sodium chloride solution and found the corrosion rate of 6.5 μm/year of steel in GGBFS blended concrete while it is the 38 μm/year in OPC concrete. Gu et. al. (2000) reported the supremacy of 55% GGBFS blended concrete with 28 days compressive strength of 45.6 MPa over OPC concrete having strength of 61.1 MPa by RCPT test. The accumulated charge passed through GGBFS blended concrete is found 670 coulombs while in normal OPC concrete, it is 1730 coulombs. Sivasundaram and Malhotra (1992) observed electrical charge ranged from 174 to 383 coulombs for various GGBFS blended concrete. Smith et. al. (2004), found that GGBFS blending benefit the initial concrete resistivity and also tend to increase the resistivity with maturity.
Zhang et. al. (1999) found that the charge passed during RCPT is 515, 775, & 675 for GGBFS blended concrete cured under moist room, 7 days burlap & then laboratory air, and with curing compounds, up to the age of testing while in OPC concrete with same curing conditions the values are 1105, 1500, & 1135 coulombs, hence they reported that the very little effect of curing conditions on chloride resistance concrete with low w/c ratio, although for all conditions the charge passed for blended concrete is lower than for OPC concrete. Detwiler et al. (1994) reported that the use of 30% slag has a far more effect on chloride resistance than lowering the w/c ratio from 0.5 to 0.4 in OPC concrete and they also found the increases in chloride diffusion with increase in curing temperature.
Sulphate Resistance
Solid Sulphate does not attack the concrete severely but when chemicals are in solution, they find the entry into porous concrete and react with the hydrated cement products. Sulphate attack of concrete takes place by the reaction of sulphate ions with Calcium hydroxide and Calcium aluminate hydrate and produced Gypsum & Ettringite with larger volume and lesser strength. Concrete with GGBFS shows improved resistance to Sulphate attack because it has lesser concentration of Calcium hydroxide and Calcium Aluminate hydrate in pore solution. Pore size and volume refinement of paste in blended concrete also improve its Sulphate resistance, but it is not a primary cause. Sulphate resistance of blended concrete depends on chemical composition of GGBFS and cement, GGBFS content in concrete, curing condition, and type of ion associated with Sulphate etc.C3A content of OPC and Alumina content of GGBFS are the primary factors related to chemical composition of binders. A replacement level of 70% or more of OPC by high Alumina slag (13- 15 %) is required for improved Sulphate resistance, where as 50 % replacement level provides good Sulphate resistance when low Alumina slag is used (Hooton and Emery, 1990). In the case of high C3A cement the 70 –80% replacement of OPC is required for better Sulphate resistance. Increase in GGBFS content in a mix increases its Sulphate resistance. Jain and Pal (1998) reported that replacement of 50% OPC by GGBFS improved the Sulphate resistance of concrete, however 70% replacement reduces the expansion of concrete significantly in Sulphate solution at the age of 6 months (Figure 7). Sulphate resistance of concrete also depends on C3S/C2S ratio (Rasheeduzzafar et. al. 1990). Low C3S/C2S ratio at a particular value of C3A increases the Sulphate resistance. Wee et. al. (2000), reported that the reduction in w/cm ratio, or increase in GGBFS content, increases the Sulphate resistance of blended concrete while change in fineness of GGBFS has no effect.
GGBFS blended concrete shows good resistance to Sodium Sulphate, but shows quick deterioration in Magnesium Sulphate attack (Figure 8). Rasheeduzzafar et. al. (1994) attributed this behavior to the depletion of Calcium hydroxide in GGBFS blended concrete. In the absence of Calcium hydroxide, Magnesium ions react more directly and extensively with C-S-H to generate noncementitious M-S-H hydrate, results in aggravated deterioration. Osborne (1999) presents the results of studies carried out at BRE (UK) and found the early curing of concrete is the most significant factor for Sulphate resistance. A beneficial effect of initial short air curing on the long-term Sulphate resistance is also reported by many researchers. This may be due to the formation of a carbonated outer layer in concrete leading to blocking of the pores and refinement of pore structures.
Aggregate-silica Reaction
Aggregate-Silica reaction (ASR) in concrete is a chemical reaction between the active silica constituents of the aggregate and the alkalies (Na+ and K+ ions) of the cement, which formed silicate gel with increase in the volume. The result is the map cracking, and disruption in concrete. Use of GGBFS in concrete reduces the risk of ASR, because of lowCa/Si ratio in the hydration products. Low Ca/Si ratio increases its alkalies binding capacity. Hence, increase in GGBFS content reduces the availability of alkalies for ASR in the concrete (Duchensne and Berude 1994) as shown in fig 9.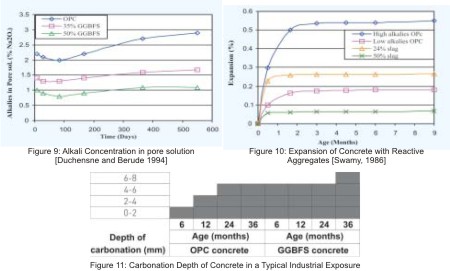
Level of GGBFS required to mitigate damaging ASR is influences by the type of reactive aggregate and the quantity of alkalies available in the mix. Expansion due to ASR is decreases with increase in GGBFS content in concrete, as shown in fig. 10 (Swamy, 1986). The 50% replacement of OPC by GGBFS is found normally satisfactory for preventing ASR with any type of aggregates and cement with less than 0.6% alkalies. As per ACI 233 report a minimum level of 40% GGBFS cement replacement is needed to mitigation of ASR.
Carbonation
In the carbonation of concrete the Carbon Dioxide (CO2) of environment reacts with hydrated compounds of cement paste and forms Calcium Carbonate. Carbonation of concrete lowers the alkalinity of pore solution and destroys the protective passive layer of reinforcement, hence increases the chances of corrosion of steel reinforcement.Carbonation resistance of GGBFS blended concrete is comparable to OPC concrete. It depends on the strength, GGBFS content and curing conditions of concrete with the environmental condition in which the concrete were situated. Concrete with 50% GGBFS achieved similar resistance to carbonation to that of OPC concrete mixture proportion, in moist environment. But higher GGBFS content (70-80%) concrete has lower resistance, especially if associated wit h a sheltered or drying climate. In dry environment concrete loss its moisture rapidly and hydration is ceases, particularly in cover concrete portion. Under these dry conditions the GGBFS blending concrete reaches lower strength and carbonated faster in earlier ages. Results of a typical field study under industrial area on the carbonation depth is shown in fig 11, suggest a marginal increase in depth of carbonation for GGBFS blended concrete.
Frost Resistance
Freezing and thawing of moisture inside the body of concrete is defined as frost action. Water gains volume (By approximately 9%) when it freezes and looses when it melts. This volume changes induce internal stresses, which do cracking, spalling, and scalling in concrete.As in OPC concrete the air content has the greatest influence on the frost resistance of GGBFS blended concrete. Frost resistance of both types concrete of similar strength and air content is essentially the same. GGBFS blended concrete without Air entraining admixture (AEA) shows significantly lower air content than in a typical OPC concrete. Dose of AEA required to entrain a given volume of air is more for GGBFS blended concrete, this may be due to the increased workability exhibited by the GGBFS blending and therefore easing the expulsion of entrapped air (Sanjayan and Sioulas, 2000). Gifford and Gillot (1996) found that air voids in GGBFS concrete is more finely divided and closely spaced than in OPC concrete with same air content. They reported that freeze thaw durability of GGBFS blended concrete is at least as good as OPC concrete given adequate air voids parameter. Due to the slower strength development at early ages in GGBFS blended concrete, its frost resistance in low temperature environment can be increase by the use of chemical activators. Deja (2003) reported that the air entraining of the concrete mix up to the level of 5-6% gives good resistance to de-icing salt in frost condition.
Conclusion
Based on the discussions presented in this paper the following conclusion have been found out:- GGBFS can be used as a high volume mineral admixture in concrete due to technological, economical, and environmental benefits, without any compromise with its performance.
- GGBFS has the cementing property, but it required activation by any medium. Hydration product of GGBFS is the same C-S-H gel with more dense and low Ca/Si ratio.
- Reactivity of GGBFS with OPC depends on the chemical and mineralogical composition of both materials. Higher fineness and glass content increases the reactivity of GGBFS. Slag Activity Index is the best way to predict the reactivity of GGBFS with OPC.
- GGBFS blending in concrete is increases the workability, consistency and reduces the water demand, bleeding & segregation chances. GGBFS blending in concrete reduces its hydration temperature and also prolonged the time for peak temperature of concrete.
- Due to the prolonged hydration of GGBFS the later age strength of GGBFS blended concrete is higher than OPC concrete under adequate reactive material and moist curing conditions.
- Chloride resistance of GGBFS blended concrete is higher than the OPC concrete due to its lower permeability and different chemistry of pore solution. Chloride resistance increases with increase in level of GGBFS in concrete.
- Sulphate resistance of concrete is increases with blending of GGBFS. Lower concentration of C3A in OPC and Al2O3 in GGBFS are favorable for better Sulphate resistance.
- Carbonation of GGBFS blended concrete is comparable to normal OPC concrete. Blended concrete exposed in long moist condition shows very little carbonation.
- GGBFS is very effective in reducing the expansion due to Aggregate-silica reaction in concrete due to higher alkalies binding capacity of hydration products of GGBFS.
- Frost resistance of GGBFS blended concrete is as good as OPC concrete, subjected to the availability of air voids. Dose of AEA required to produce same air content is higher in blended concrete.
- To get complete advantages of GGBFS blending in concrete minimum 7 day moist curing is must. Replacement level of 40 to 50% is the optimum from strength and durability aspects.
References
- ACI 233R-95 Committee Report, (1997), GGBFS as a Cementitious Constituents in Concrete, ACI Manual of Concrete Practice, Part I
- Al-Amoudi, O. S. B., Rasheeduzzafar, Maslehuddin, M., and Al-Mana, A. I., (1993), Prediction of Long-term Corrosion Resistance of Plain and Blended Cement Concrete, ACI Materials Journal, Vol. 90, No. 6, pp. 564-570
- Babu, K. G., and Kumar, V. S. R., (2000), Efficiency of GGBFS in Concrete, Cement and Concrete Research, Vol. 30, pp. 1031- 1036
- Bijen, J., (1996), Benefits of Slag and Fly Ash, Construction and Building Materials, Vol. 10, No. 5, pp. 309-314
- Brooks, J.J., and Al-Kaisi, A. F., (1990), Early Strength Development of Portland and Slag Cement Concrete Cured at Elevated Temperatures, ACI Materials Journal, Vol. 87, No. 5, pp. 503-507
- Deja, J., (2003), Freezing and De-Icing Salt Resistance of Blast Furnace Slag Concretes, Cement and Concrete Composites, Vol. 25, pp. 357-361
- Detwiler, R. J., Fapohunda, C. A., and Natale, J., (1994), Use of Supplementary Cementing Materials to increase the Resistance to Chloride ion Penetration of Concrete Cured at Elevated temperatures, ACI Materials Journal, Vol. 91, No. 1, pp. 63-66
- Duchesne, J., and Berube, M. A., (1994), Available Alkalies from Supplementary Cementing Materials, ACI Materials Journal, Vol. 91, No. 3, pp. 289-299
- Garcia, J. I. E., Fuentes, A. F., Gorokhovsky, A., Fraire-Luna, P. E., and Mendoza-Suarez, G., (2003), Hydration Products and Reactivity of Blast-Furnace Slag Activated by various Alkalis, Journal of American Ceramic Society, Vol. 86, No. 12, pp. 2148-2153
- Geiseler, J., Kollo, H., and Lang, E., (1995), Influence of blast furnace cements on durability of concrete structures, ACI Materials Journal, Vol. 92, No. 3, pp. 252- 257
- Gifford, P. M., and Gillott, J.E., (1996), Freeze-Thaw Durability of Activated Blast Furnace Slag Cement Concrete, ACI Materials Journal, Vol. 93, No. 3, pp. 242- 245
- Gu, P., Beaudoin, J. J., Zhang, M. H., and Malhotra, V. M., (2000), Performance of Reinforcing Steel in Concrete Containing Silica Fume and Blast- Furnace Slag Ponded with Sodium Chloride Solution, ACI Materials Journal, Vol. 97, No. 3, pp. 254-262
- Hooton, R. D., and Emery, J. J., (1990), Sulphate Resistance of a Canadian Slag Cement, ACI Materials Journal, Vol. 87, No. 6, pp. 547-555
- Hope, B. B., and Lp, A. K. C., (1987), Corrosion of Steel in Concrete made with Slag Cement, ACI Materials Journal, Nov.- Dec., 525-531
- Jain, M. K., and Pal, S. C., (1998), Utilisation of industrial slag in making high performance concrete composites, Indian Concrete Journal, June, pp. 307- 315
- Jimenez, A. F., and Puertas, F., (2003), Structure of Calcium Silicate Hydrates Formed in Alkaline-Activated Slag: Influence of the Type of Alkaline Activator, Journal of American Ceramic Society, Vol. 86, No. 8, pp. 1389-1394
- Kumar, S., Rao, B. K., and Mishra, S., (2002), Chloride Penetration Resistance of Concrete Containing Blast Furnace Slag, The Indian Concrete Journal, December, pp. 745-751
- Lim, S. N., and Wee, T. H., (2000), Autogeneous Shrinkage of Ground- Granulated Blast-Furnace Slag Concrete, ACI Materials Journal, Vol. 97, No. 5, pp. 587-593
- Mantel, D. G., (1994), Investigation into the Hydraulic Activity of Five Granulated Blast Furnace Slag with Eight Different Portland Cements, ACI Materials Journal, Vol. 91, No. 5, pp. 471-477
- Miura, T., and Iwaki, I., (2000), Strength Development of Concrete Incorporating High level of ground Granulated Blast- Furnace Slag at Low Temperatures, ACI Materials Journal, Vol. 97, No. 1, pp. 66- 70
- Olorunsogo, F. T., and Wainwright, P. J., (1998), Effect of GGBFS Particle-Size Distribution on Mortar Compressive Strength, A.S.C.E. Journal of Materials in Civil Engineering, August, pp. 180-187
- Osborne, G. J., (1999), Durability of Portland Blast-Furnace Slag Cement Concrete, Cement and Concrete Composites, Vol. 21, pp. 11-21
- Rasheeduzzafar, Al-Amoudi, O. S. B., Abduljauwad, S. N., and Maslehuddin, M., (1994), Magnesium-Sodium Sulphate attack in Plain and Blended Cements, A.S.C.E. Journal of Materials in Civil Engineering, Vol. 6, No. 2, pp. 201-221
- Rasheeduzzafar, Dakhil, F. H., Al-Gahtani, A. S., Al-Saadoun, S. S., and Bader, M. A., (1990), Influence of Cement Composition on the Corrosion of Reinforcement and Sulphate Resistance of Concrete, ACI Materials Journal, Vol. 87, No. 2, pp. 114- 122
- Roy, D. M., and Idorn, G. M., (1982), Hydration, Structure, and Properties of Blast Furnace Slag Cements, Mortar, and Concrete, ACI Materials Journal, Nov.– Dec., pp. 444-457
- Sanjayan, J. G., and Sioulas, B., (2000), Strength of Slag-Cement Concrete Cured in place and in other conditions, ACI Materials Journal, Vol. 97, No. 5, pp. 603- 611
- Sivasundaram, V., and Malhotra, V. M., (1992), Properties of Concrete Incorporating Low Quantity of Cement and High Volumes of Ground Granulated Slag, ACI Materials Journal, Vol. 89, No. 6, pp. 554-563
- Smith, K. M., Schokker, A. J., and Tikalsky, P. J., (2004), Performance of Supplementary Cementitious Materials in Concrete Resistivity and Corrosion Monitoring Evaluations, ACI Materials Journal, Vol. 101, No. 5, pp. 385-390
- Swamy, R. N., (1986), Cement replacement Materials, Surrey University Press, London
- Swamy, R. N., and Bouikini, A., (1990), Some Engineering Properties of Slag Concrete as Influenced by Mix Proportioning and Curing, ACI Materials Journal, Vol. 87, No. 3, pp. 210-220
- Wee, T. H., Suryavanshi, A. K., and Tin, S. S., (2000), Evaluation of Rapid Chloride Permeability Test, Results for Concrete Containing Mineral Admixtures, ACI Materials Journal, Vol. 97, No. 2, pp. 221- 23
Published on:
08 January 2009
Published in: NBM&CW November 2007
Share:
We Value Your Comment




































