Ferro-Sialate Geopolymer using Red Mud
Geopolymer is being used as a replacement for Portland cement for over the past three decades. The technology utilizes material rich in silica and alumina, either processed geological material or industrial byproducts as the raw material for the binder synthesis in the presence of alkaline medium. Natural alumino-silicate minerals used are mostly kaolin and metakaolin, whereas fly ash, slag, waste glass and red mud are the common industrial byproducts used for geopolymerisation. Agricultural byproducts like rice husk ash and palm oil fuel ash are also being used to synthesize geopolymer products. The properties of the geopolymer products vary with the type of raw material used. Apart from mitigating carbon emission, the greatest advantage of using the geopolymer technology is its capability to immobilize and encapsulate hazardous materials that become a part of the geopolymer matrix.
Prior to year 2010, there was the general opinion that the presence of iron was detrimental to the strength development of geopolymer. In 2008, Davidovits [1] recommended the percentage of Fe2O3 to be less than 10% and suggested that the presence of hematite and magnetite greatly decreased the compressive strength of geopolymer. In 2010, C. K. Gomes et al. [2] studied geopolymerisation of iron-rich lateritic soil containing 60% of iron. They reported the presence of Fe-O bonds in the geopolymer matrix. Mössbauer spectra analysis revealed the presence of structural Fe3+ inside an aluminosilicate structure, which suggested the replacement of Al3+ by Fe3+ in octahedral sites.
In his keynote presentation at geopolymer camp, 2012, Davidovits [3] introduced a new type of geopolymer binder called ferro-sialate-based geopolymer cement and also filed a patent for the same. The novel binder possessed the same properties as rock-based geopolymer binder but comprised of geological elements rich in iron content. The structure was of type (Ca, K, Na)-(-Fe-O)-(Si-O-Al-O-), fig. 1. Ferro-sialate-based geopolymer remains the focus of research in geopolymerisation field and has not been commercialised yet.
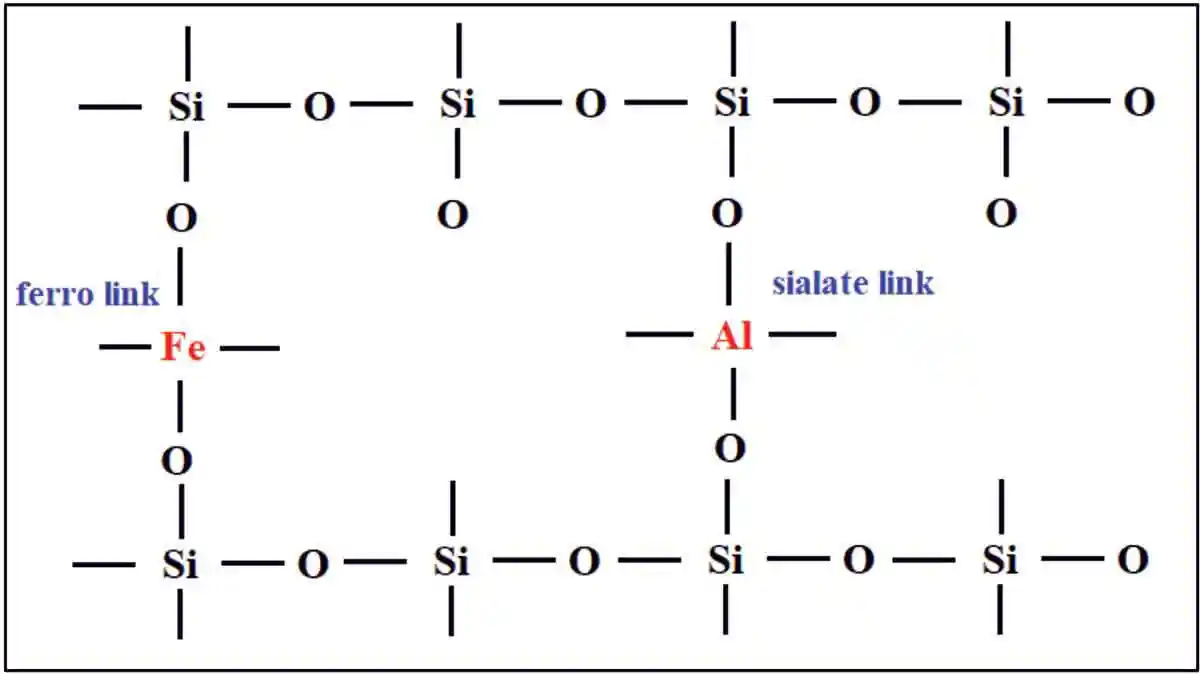 Fig. 1 Ferro-sialate geopolymer structure
Fig. 1 Ferro-sialate geopolymer structureIn 2014, Esther A. Obonyo et al. [4] carried out investigation on geopolymerisation of two types of laterite soil, the first one with 9.68% of Fe2O3 and the second one containing 35.2% of Fe2O3. Kaolinite was the prominent mineral in both the type of soils. He studied the implications of incorporation of Fe3+ ion in laterite-based geopolymer matrix. As reported by him, iron played a key role during the geopolymerisation of the lateritic by creating a state of disorder in the structure of kaolinite, making it more vulnerable for dissolution and thereby assisting the geopolymerisation process. In the present investigation, the geopolymerisation of red mud is studied which is an iron-rich aluminosilicate material.
Material Characterisation
Red mud is a solid waste produced during the extraction of alumina from bauxite by Bayer’s process. Approximately, 1.5 to 2.5 tons of red mud is generated per ton of alumina produced depending on the raw material processed. The generation of red mud in India is reported to be 4.71 million tons, which is 6.7% of the global production of red mud, which is around 70 million tons [5]. There is continuous and rapid increase in the inventory of red mud. Its disposal remains a major concern for aluminum plants. Red mud cannot be used alone as a raw material for geopolymerisation due to its extreme fineness and cohesiveness, hence it was mixed with fly ash in varying proportion. Slag was used at constant 10% for all variations in order to facilitate ambient curing. Table 1 shows the chemical composition of red mud, fly ash and GGBS used. Iron is a predominant constituent of red mud comprising of nearly 40% of it.The red mud obtained was coarsely ground and passes through 300 micron sieve to ensure uniformity in test results.
| Table 1 Chemical composition of red mud, fly ash and GGBS | |||
| Metal Oxide% | RM | FA | GGBS |
| SiO2 | 9.93 | 61.54 | 37.73 |
| Al2O3 | 18.1 | 25.37 | 14.42 |
| Fe2O3 | 42.9 | 6.73 | 1.11 |
| CaO | 2.3 | 3.1 | 37.34 |
| K2O | - | - | - |
| Na2O | 5.58 | 0.97 | - |
| MgO | - | 0.73 | 8.71 |
| TiO2 | 9.03 | - | - |
| P2O5 | 0.35 | - | - |
| V2O5 | 0.31 | - | - |
| SO3 | - | 0.62 | 0.39 |
| L.O.I | 10.5 | 0.39 | 1.41 |
The gradation curve showed that 90% of the red mud samples were found to be finer than 75μm and 80% were finer than 45μm. SEM image as shown in fig. 2. The silica content of red mud mostly is unreactive and exists in crystalline state as reflected in mineralogical analysis in fig.3. Lime reactivity of red mud was low with an average value of 0.95 MPa,which indicated its poor pozzolanicbehaviour. Silica reactivity test conducted on red mud samples, indicated the average percentage of reactive silica to be 1.6% only.Mechanical activation of red mud was done in order to improve its reactivity.
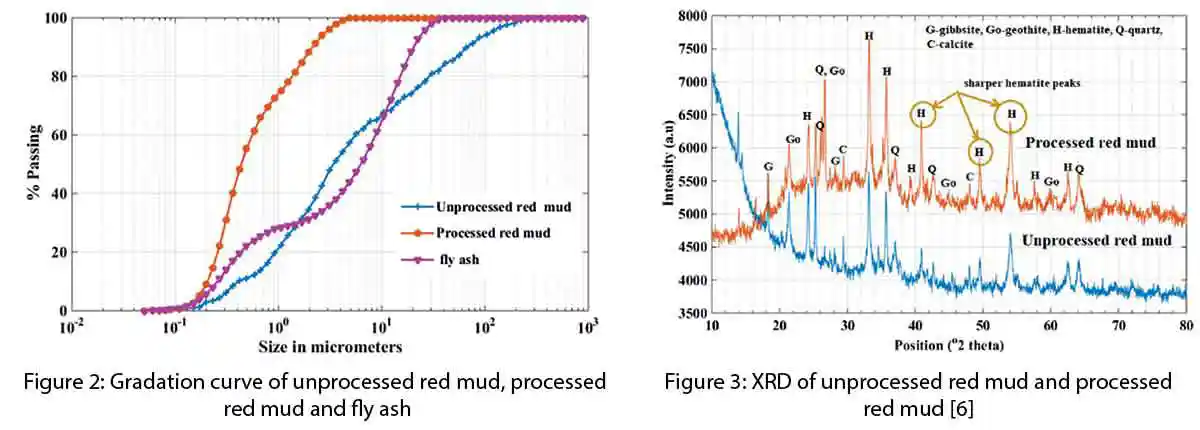
The processing of red mud increased its reactivity with lime from 0.95 MPa to 3.61 MPa, which was a significant increment of more than 300%. Grinding of red mud reduced the particle size and probably activated some of the silica that was otherwise crystalline to react with lime-forming di-calcium silicate. Silica reactivity test affirmed the increase in reactive silica percentage from 1.62% to 5.12%. Reactive silica of fly ash was 27.12% but its lime reactivity was lesser than that of pulverized red mud, this hinted at the participation of iron in increasing the reactivity of red mud. The XRD pattern of pattern of pulverised red mud and fig. 3 compares the diffraction pattern of both unprocessed and pulverised red mud. There was no major change observed in the diffraction pattern of both red mud samples but the hematite peaks were observed to be sharper. The SEM image of processed red mud in fig.4 no longer showed big lumps of particles. Grinding of red mud led to the dispersion of bigger agglomerates of hematite into nano-sized particles, fig. 5. Pulverisationof red mud presumably not only activated some silica for geopolymerisation but also made hematite more reactive.
 Figure 4: SEM of unprocessed red mud and processed red mud
Figure 4: SEM of unprocessed red mud and processed red mud![TEM of a) unprocessed and b) processed red mud [7].](/images/87-Concrete/41778-Fig-5.webp) Figure 5: TEM of a) unprocessed and b) processed red mud [7].
Figure 5: TEM of a) unprocessed and b) processed red mud [7].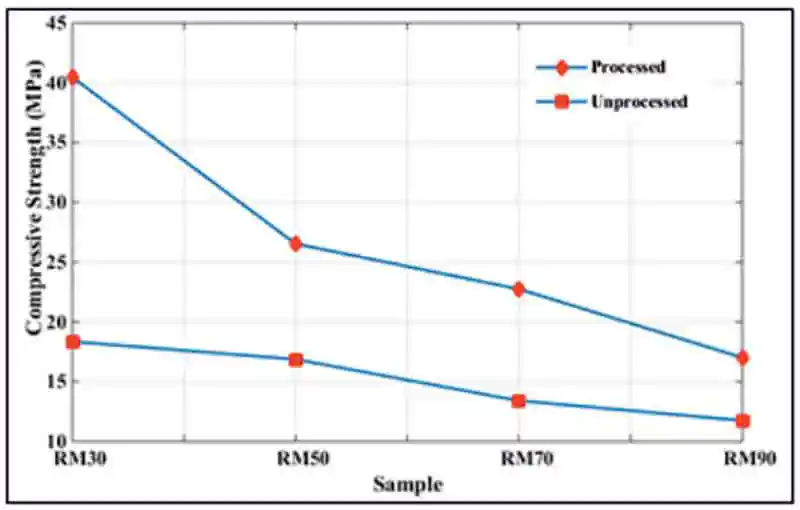 Figure 6: Compressive strength of processed and unprocessed red mud geopolymer
Figure 6: Compressive strength of processed and unprocessed red mud geopolymerIt was iron in red mud that was making the difference as the alumina content of fly ash exceeded that of red mud. Past investigations have proven the existence of Fe-O bond in the geopolymeric structure [2, 3]. Furthermore, Esther A. Obonyo et al. [4] suggested that the presence of iron enhanced the dissolution rate of silica and alumina of kaolinite in laterite. In the present investigation, iron seemed to have not only participated in geopolymerisation but, more importantly, indirectly contributed to geopolymerisation by activating some other phases.
In order to validate the contribution of iron in geopolymerisation, Mössbauer Spectroscopy test was carried out. The Mössbauer spectral parameter was fitted using “NORMOS” computer programme. Fig. 7 shows the fitted Mössbauer spectra of unprocessed red mud (RM), pulverised red mud (PRM), RM30 and PRM30 samples at room temperature. As shown in the fig., the Mössbauer spectrum of each sample is fitted with two sub spectrum; one sextet and one doublet. The Mössbauer fitting parameters are tabulated in table 2.
| Table 2.Mössbauer spectral fitting parameters of RM, PRM, RM30 and PRM30 sample. BHf = Hyperfine field, I.S = Isomer shift, Q.S = Quadruple splitting, ∆ = Width of spectra | ||||||
| Sample code | Sub-spectrum | BHf(T) | I.S (mm/s) | Q.S (mm/s) | ∆ (mm/s) | Area (%) |
| RM | S1 | 50.40 | 0.264 | -0.214 | 0.493 | 61.47 |
| D1 | 0.250 | 0.598 | 0.450 | 38.53 | ||
| PRM | S1 | 50.51 | 0.255 | -0.250 | 0.428 | 63.73 |
| D1 | 0.260 | 0.578 | 0.424 | 36.27 | ||
| GRM30 | S1 | 50.49 | 0.263 | -0.202 | 0.511 | 60.22 |
| D1 | 0.255 | 0.587 | 0.478 | 39.78 | ||
| GPRM30 | S1 | 50.58 | 0.265 | -0.233 | 0.497 | 63.09 |
| D1 | 0.243 | 0.589 | 0.462 | 36.91 | ||
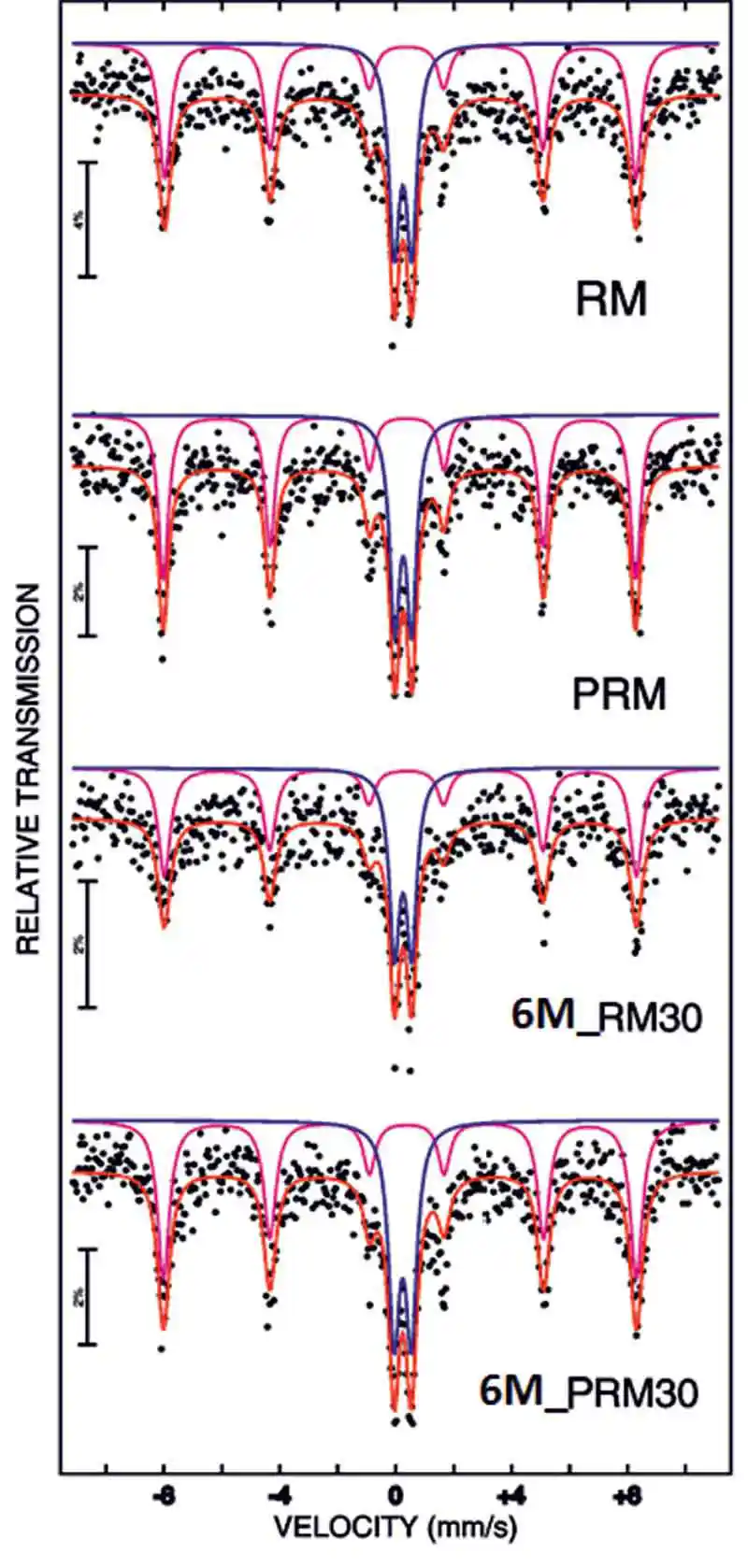 Figure 7: Room temperature Mössbauer spectra of RM, PRM, 6M_RM30 and 6M_PRM30 sample. The data points are shown in black dots
Figure 7: Room temperature Mössbauer spectra of RM, PRM, 6M_RM30 and 6M_PRM30 sample. The data points are shown in black dotsPulverisation of red mud would have caused the partial transformation of paramagnetic ferrihydrite phases into anti-ferromagnetic hematite phase. This was evident from the increase in percentage relative area of sextet of pulverised red mud sample (PRM).
From the table 2, it is observed that polymerisation of red mud and pulverised red mud has led to increase in the area percentage of doublets. This increase in area percentage might be due to replacement of aluminium with iron in the geopolymerised sample (RM30 and GRM30). As we know that geopolymer is an inorganic polymer with silicate and aluminate linkages (Si-O-Si and Si-O-Al). Gepolymerisation of red mud would cause the partial replacement of Al3+ ions with Fe3+ ions into the alumina-silicate linkage. The presence of Fe3+ in the silicate linkage in the paramagnetic form would result in increment of the doublet in Mössbauer spectra.
Conclusion
A large inventory of red mud is present globally which could be hazardous if left to pile up. The main constituent of red mud is iron oxide and it has a potential to be used in geopolymerisation due to presence of alkalies, silica and alumina. Results presented in the characterization test on red mud show that it is non pozzolanic in nature due to the prevalence of non - reactive silica. But the existence of agglomerates of red mud particles in nano scale range and its high alkalinity is likely to have a favourable influence on geopolymerisation process and strength development. Pulverization reduced the particle size and increased the fineness and reactive silica of red mud.
Mössbauer spectroscopy and FTIR tests on the geopolymerised binder confirmed the participation of hematite phase of red mud in geopolymerisation. Pulverisation of red mud not only enhanced its pozzolanic behavior by increasing the percentage of reactive silica but also increased the reactivity of the hematite phase. The recommended use of ambient cured red mud based geopolymer could be in the production of high strength concrete, bricks/blocks and pavers. Ferro-sialate based GPC has a great potential in India due to the presence of huge deposits of unutilized mine wastes.
References
- J.Davidovits, “Geopolymer Chemistry & Applications”, Geopolymer Institute, 2008, Chapter 12.
- G.K.Cristiane, G.S.T.Lima, T.S.Marden, De Barros Silvio, V.I.Frota, B. N.Perazzo, “Iron distribution in geopolymer with ferromagnetic rich precursor”, Materials Science Forum Vol. 643,2010, pp 131-138.
- J.Davidovits,“State of the Geopolymer R&D”, 2012. http://www.geopolymer.org/camp/gpcamp-2012.
- A.EstherObonyo, E.Kamseu,P.N. Lemou- gna, B.Arlin,Tchamba, U.C. Melo and C.Leonelli, “A Sustainable Approach for the Geopolymerization of Natural Iron-Rich Aluminosilicate Materials”, Sustainability, Vol. 6, 2014, pp 5535-5553.
- G.N.Reddy and K.S.Chandra, “Characteri- zation and Comprehensive Utilization of Red mud - An Overview”, International Journal for Scientific Research & Development, Vol. 2, Issue 1, 2014, pp 2321-0613.
- S. Singh, M.U. Aswath, and R. V. Ranganath, Effect of mechanical activation of red mud on the strength of geopolymer binder, Constr. Build. Mater. 177 (2018) 91–101. doi:10.1016/j.conbuildmat.2018.05.096.
- Singh S, Aswath MU, Das Biswas R, et al (2019) Role of iron in the enhanced reactivity of pulverized Red mud: Analysis by Mössbauer spectroscopy and FTIR spectroscopy. Case Stud Constr Mater 11:e00266. https://doi.org/https://doi.org/10.1016/j.cscm.2019.e00266
- W.Salama, M.El Aref, Reinhard Gaupp, “Spectroscopic characterization of iron ores formed in different geological environments using FTIR, XPS, Mössbauer spectroscopy and thermoanalyses”, Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, Vol. 136, 2015, pp 1816-1826.
- U.Schwertmann, E.Murad, “Clays”, Clay Minerals, Vol. 31, 1983, pp 277.
- U.Schwertmann, J. Friedl, H. Stanjek, Journal of Colloid and Interface Science, Vol. 209, 1999, pp 215-223.
- U.Schwertmann, H.Stanjek, H.H. Becher, Clay Miner., Vol. 39, 2004, pp 433-438.
- Y.Cudennec, A. Lecerf, “The transformation of ferrihydrite into goethite or hematite, revisited”. J.Solid State Chemistry. Vol. 179 (3), 2006 pp 716–722.
- D.B.Hausner, N.Bhandari, Andro-Marc Pierre-Louisa, J.D. Kubicki, D.R. Strongin, “Ferrihydrite reactivity toward carbon dioxide”, Journal of Colloid and Interface Science, Vol. 337, 2009, pp 492–500.

He is the Bureau Head Bangalore for SME World, New Delhi, Technical Adviser for RMCMA and former Secretary General for All India ACCE (I) and former Vice president (south) of Indian Concrete Institute. As Training and Placement officer, he has placed more than 35 thousand students in national and multinational companies. He joined Rotary in the year 2009 and took up the Presidentship in 2012-13 during which time the club took a new shape and initiated major events like SME National Awards for Excellence, Spell Star, NufACE etc. He was President of Rotary Bangalore south east and Assistant Governor in Rotary International district 3190.
He has published more than 100 papers in national and international journals/conferences and has organinised more than 300 workshops/seminars for professionals/faculty members/students for knowledge dissemination. His list of awards includes:
- “Achievement Award for Outstanding Academician”, CIDC Vishwakarma Award 2014, The National Award is conferred on 7th March 2014 by Construction Industry Development Council (CIDC) established by Planning Commission, Government of India and the Construction Industry.
- Felicitation by the Honourable Chief Minister-GoK, Shri. Jagadeesh Shettar for editing the Manual: Editor-in-Chief for “Reference Manual for Field Engineers on Building Construction” published by Task Force for Quality Assurance in Public Constructions, Government of Karnataka.
- Eminent Engineer By ACCE(I) Bangalore Centre, Felicitation during the Engineers Day Celebration, September 2011
- Academician Par Excellence by Rotary


Published on:
19 May 2020
Published in: NBM&CW May 2020
Share:
We Value Your Comment




































